EXTERNAL BEAM RADIOTHERAPY
The goal of radiotherapy is to deliver a high dose of irradiation on a target volume somewhere in the human body while keeping the dose given to other parts or organs of the body to a minimum. The high radiation dose will then cure, or help to cure the disease, in most cases a tumour. If the tumour cannot be cured, the radiation is expected to slow down the tumour progression and to decrease symptoms induced by the tumour.
Different techniques are available to obtain the required radiation dose in the target area. In this paragraph, we will concentrate on the most frequently used radiotherapy techniques based on the use of a machine that sends irradiation to the body without touching the body: External beam radiotherapy.
Modern external radiotherapy techniques allow for a high dose to be given very precisely. This translates to higher tumour shrinkage and lowered side-effects. It is impossible to explain the exact effects here because this depends highly on the type of tumour and/or the location in the human body.
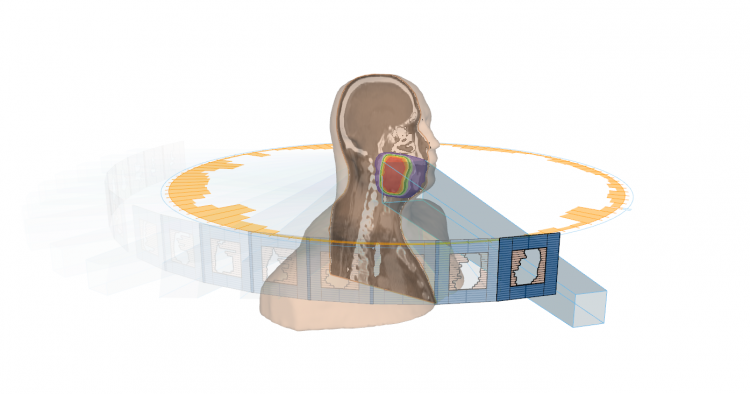
The goal of the treatment and different approaches
- Curative (radiotherapy alone)
- Curative (as an adjuvant therapy to surgery)
- Quality of life-improving
Curative (radiotherapy alone)
The goal of this type of external beam radiotherapy is to deliver a dose that is high enough to make a tumour disappear, without the need for surgery. Because of the high dose, this type of therapy is not always possible in case of large tumours, or tumours that are not sensitive enough to radiotherapy. To increase the dose, this treatment can sometimes be delivered using stereotactic techniques: see further down this page.
Curative (as an adjuvant therapy to surgery)
A common situation is the use of surgery to remove the main tumour, if possible also with adjoining tissue or lymph node areas where the tumour might have invaded in or spread to. However, there can be a risk of tumour recurrence. This means that for some tumours, apparently, the surgery did not manage to eradicate all of the (microscopically small) disease. The addition of an external beam radiotherapy treatment to the whole operated area is then often used to eradicate the remaining tumour cells. Because a larger area is usually treated, the dose is high but not as high as in the previous paragraph. This means that the radiation will only cure small remaining groups of tumour cells. In the event of a higher risk of remaining tumour cells, it can be useful to increase very focally the dose of irradiation. This is called a 'boost'.
Quality of life-improving
Surgery, systemic treatment, medication and radiotherapy cannot obtain a real and definite cure for every patient. The life expectancy of the patient can then be threatened by the tumour. In cases like this, the goal of the medical team will be to help the patient to live a life as comfortable as possible. Medical treatments, including radiotherapy, no longer have a curative goal, but they are very important because they can prevent or treat problems like pain, compression, bleeding, etc. Even if this was not the primary goal, there are sometimes even beneficial effects on the duration of life.
There was a time that radiotherapy treatments were either'very curative' or 'very palliative'. But with the help of better systemic treatments and more efficient radiotherapy, there is now a large group of patients who benefit from a treatment that is not strictly curative, but still very different and much more effective than the old 'simple' palliative treatments. That is why we like speaking of 'quality of life improving treatments', avoiding a bit the possibly misunderstood term 'palliative'.
An external beam radiotherapy treatment is done in multiple steps
- Multidisciplinary discussion
- Consultation
- Paramedical help
- Simulation
- Supplementary imaging (in some cases)
- Concomitant chemotherapy (in some cases)
- Treatment planning
- First treatment, with imaging
- Quality control during the treatment
- Further treatment sessions
- Consultations during the treatment
- Consultation after the treatment
Multidisciplinary discussion
After the initial diagnostic procedures, the data concerning the tumour and the patient are discussed in the presence of physicians from different specialties. The general physician and other healthcare team-members might also be present. A treatment strategy is then developed.
Consultation
A physician, usually having participated in the multidisciplinary discussion, sees the patient during an initial (longer) consultation, to explain the proposed treatment strategy. For the radiotherapy part, the technical modalities of this treatment will be explained. Information on desired and undesirable effects will be given. Patients can sometimes be invited to participate in medical trials.
Paramedical help
Cancer can induce a number of problems beyond the tumour location such as mobility reduction, pain and nutritional problems. The disease can also influence the functioning of the patient in his/her family, workplace and society in general. While a medical treatment is of prime importance, the radiotherapy team wants the patient to be well assisted in tackling other problems as well. Therefore most radiotherapy departments will be able to bring the patient into contact with social support teams, pain clinics, dieticians, massage therapists, ergonomists, psychologists and more.
Simulation
The radiotherapy treatment will be calculated based on images obtained during the simulation. This is a preparatory procedure during which the patient is positioned on the simulator table in exactly the same way as on the real treatment machine. Immobilisation material is applied, and a set of 3D images (CT scans) is obtained. Reference points are marked on the skin. The used immobilisation material might be made to measure.
Supplementary imaging (in some cases)
The simulator CT scan may not show with enough detail the exact location of the tumour or certain organs to be avoided for each patient. A supplementary MRI or PET-CT scan can then be obtained, ideally with the patient placed in the same position as will be the case during the real treatment. The advantage of the PET-CT scan is the availability of 'metabolic information', which means it is possible to visualize the areas where the tumour is most active. New PET-CT scan techniques can also document other behaviours of the tumour: where is it growing faster? Where is it lacking oxygen? It is expected that this information will help to even better adapt the radiation dose to the tumour.
Concomitant chemotherapy (in some cases)
The risk of spreading of the tumour can be reduced by a systemic treatment, like chemotherapy. Chemotherapy can be given before or after the radiotherapy treatment. There is, however, a third option: to add the chemotherapy during the radiotherapy treatment. This might cause the chemotherapy to increase the effect of the radiotherapy and is called the 'radio-sensitising effect of chemotherapy'.
Treatment planning
Once the patient is positioned on the table, the immobilisation material that was made or used during the simulation will be applied. Then a verification of the correct position of the patient is done. A portal image or cone beam CT is made, helping to verify this positioning. In case of small deviations, the table and/or the patient are repositioned. The actual treatment can now start. The time of irradiation is often less than 10 minutes, but including the imaging, the duration the patient will reside in the treatment vault (bunker) can be a bit longer.
First treatment, with imaging
After being positioned on the table, the immobilisation material will be applied. The patient is then ready for a first technical control to verify if (s)he is indeed correctly placed: a portal image or cone beam CT is made, helping to verify this positioning. In case of small deviations, the table and/or the patient are repositioned. Then the treatment itself can start. The irradiation itself often takes less than 10 minutes, but including the imaging the duration the patient will reside in the treatment vault (bunker) can be a bit longer.
Quality control during the treatment
Portal imaging and cone beam CTs are not the only ways to verify the correct execution of the treatment, other measurements can be done as well. For instance: measuring which dose is detected near the skin at the entrance of a treatment beam, or the dose still found at the exit of the beam(s).
Further treatment sessions
A treatment session is also called a fraction. There can be anywhere between 1 and 40 fractions, often one per workday, but other variations exist. As treatment continues, the dose given to the tumour gradually accumulates. Treatment techniques are sometimes modified in the course of the treatment. This may be planned in advance or might be decided during an on-going treatment.
Consultations during the treatment
Every few days the patient will have the opportunity to encounter a member of the (para)medical team to discuss treatment progress and tolerance to the irradiation. If required, medication or other measures can be proposed to treat possible side effects.
Consultation after the treatment
At the end of the treatment, the patient will again meet with the radiotherapy team to verify if any short-term side effects are present and to discuss how the further follow-up will be organised.
Different techniques of external beam radiotherapy
Simple irradiation techniques
Irradiation with one single or two beams is only used in areas or situations that are very easy to treat, such as:
- A direct beam with 'electrons' on a superficially located target volume;
- A very simple treatment with 2 beams in a patient who is too weak to support or justify a longer or more complex treatment.
This approach is rapid, simple and happens at a low cost.
3-dimensional conformal therapy
This is a more precise approach. The target volume is identified on the computer screen (showing the CT scan made during the simulation). The treatment is then calculated, based on sending a combination of 3 to 4, 5, 6, 7 beams to the target volume. The size, energy and form of these beams are adapted to the location and form of the target volume.
IMRT
Intensity-modulated radiotherapy (IMRT) is a further refining of the conformal radiotherapy. With IMRT not just the whole beam is adapted in form and intensity, but different 'sub-beams' can each have their own form and intensity. This allows for a better sculpting of the dose around the target volume, reducing inhomogeneities induced by the bizarre form of the tumour and the presence of inhomogeneities in the line of the beams (cavities, dense bony structures, ...). This technique helps to avoid small areas with a relatively too high dose (near organs at risk) or too low dose (in the tumour), reducing side effects and increasing cure rates.
Rotational IMRT
In 3D Conformal Therapy the daily doses is distributed over a number of beams whereby each fraction is given in one go. In ‘classic’ IMRT, static beams are used whereby the treatment is interrupted at several stages in order to irradiate each part of the target volume. This technique is called ‘step and shoot’ and it paints the target volume in such a way that an overdoses or an underdoses is avoided. Increasing the number of beams results in an increased equal spreading of the dose distribution. The highest number of beams is achieved by giving the irradiation while the treatment machine turns around the patient in a 360° movement. A number of different machines have this capability: RapidArc or VMAT technology, TomoTherapy, Novalis, CyberKnife.
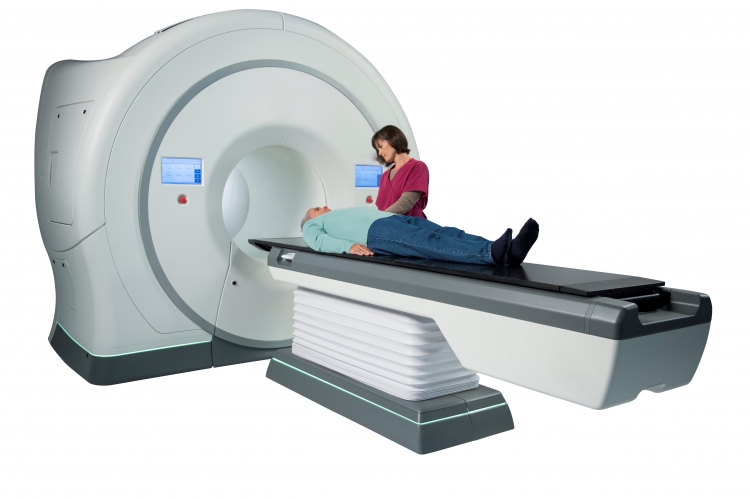
Image-guided radiotherapy (IGRT)
Highly precise treatment plans can only be executed correctly when the target volume is placed in the isocenter of the beam with great precision. In order to achieve this, images of the area that needs to be irradiated are taken. These images are used to correct potential inaccurate patient positions before the actual irradiation treatment is administered.
